Hard water and water softening
Every household and every factory uses water, and none of it is pure. One class of impurity that is of special interest is "hardness". This refers to the presence of dissolved ions, mainly of calcium Ca2+ and magnesium Mg2+ which are acquired through contact with rocks and sediments in the environment. The positive electrical charges of these ions are balanced by the presence of anions (negative ions), of which bicarbonate HCO3– and carbonate CO32– are most important. These ions have their origins in limestone sediments and also from carbon dioxide which is present in all waters exposed to the atmosphere and especially in groundwaters.
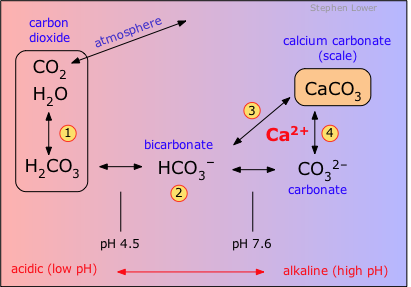 |
Origin of water "hardness" Carbon dioxide reacts with water to form carbonic acid (1) which at ordinary environmental pH exists mostly as bicarbonate ion (2). Microscopic marine organisms take this up as carbonate (4) to form calcite skeletons which, over millions of years, have built up extensive limestone deposits. Groundwaters, made slightly acidic by CO2 (both that absorbed from the air and from the respiration of soil bacteria) dissolve the limestone (3), thereby acquiring calcium and bicarbonate ions and becoming "hard". If the HCO3– concentration is sufficiently great, the combination of processes (2) and (4) causes calcium carbonate ("lime scale") to precipitate out on surfaces such as the insides of pipes. (Calcium bicarbonate itself does not form a solid, but always precipitates as CaCO3.) |
 These "hardness ions" cause two major kinds of problems. First, the metal cations react with soaps, causing them to form an unsightly precipitate— the familiar "bathtub ring".
These "hardness ions" cause two major kinds of problems. First, the metal cations react with soaps, causing them to form an unsightly precipitate— the familiar "bathtub ring".
 More seriously, the calcium and magnesium carbonates tend to precipitate out as adherent solids on the surfaces of pipes and especially on the hot heat exchanger surfaces of boilers. The resulting scale buildup can impede water flow in pipes. In boilers, the deposits act as thermal insulation that impedes the flow of heat into the water; this not only reduces heating efficiency, but allows the metal to overheat, which in pressurized systems can lead to catastrophic failure.
More seriously, the calcium and magnesium carbonates tend to precipitate out as adherent solids on the surfaces of pipes and especially on the hot heat exchanger surfaces of boilers. The resulting scale buildup can impede water flow in pipes. In boilers, the deposits act as thermal insulation that impedes the flow of heat into the water; this not only reduces heating efficiency, but allows the metal to overheat, which in pressurized systems can lead to catastrophic failure.

Bad Hair Day? "Hard water hair" can be avoided by rinsing with a mildly acidic solution which will dissolve the carbonate deposits. Try adding some vinegar, lemon juice or citric acid to water.
Types of water hardness
Temporary hardness
This refers to hardness whose effects can be removed by boiling the water in an open container. Such waters have usually percolated though limestone formations and contain bicarbonate HCO3– along with small amounts of carbonate CO32– as the principal negative ions. Boiling the water promotes the reaction
2 HCO3– → CO32– + CO2 + H2O
by driving off the carbon dioxide gas. The CO32– reacts with Ca2+ or Mg2+ ions, to form insoluble calcium and magnesium carbonates which precipitate out. By tying up the metal ions in this way, the amounts available to form soap scum are greatly reduced.
Permanent hardness
Waters than contain other anions such as chloride or sulfate cannot be remediated by boiling, and are said to be "permanently" hard. The only practical treatment is to remove all the ions, normally by the method described below.
For more about the classification of hard waters, see this excellent Wikipedia article.
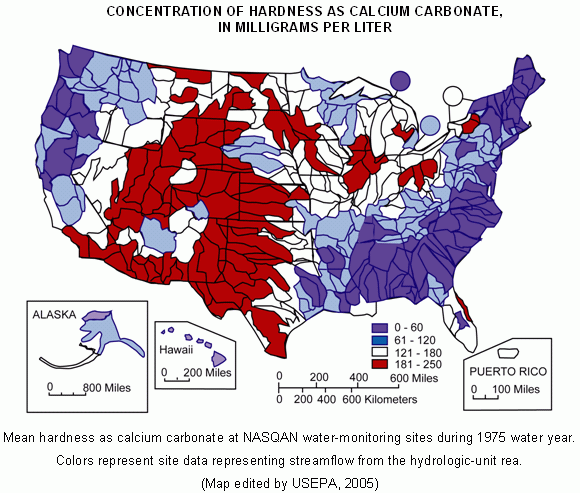
The following map from the USGS shows how mean water hardness varies in the U.S.
Conventional water softening
Most conventional water-softening devices depend on a process known as ion-exchange in which "hardness" ions trade places with sodium and chloride ions that are loosely bound to an ion-exchange resin or a zeolite (many zeolite minerals occur in nature, but specialized ones are often made artificially.)

The illustration depicts a negatively-charged zeolite to which [positive] sodium ions are attached. Calcium or magnesium ions in the water displace sodium ions, which are released into the water. In a similar way, positively-charged zeolites bind negatively-charged chloride ions (Cl–), which get displaced by bicarbonate ions in the water. As the zeolites become converted to their Ca2+ and HCO3– forms they gradually lose their effectiveness and must be regenerated. This is accomplished by passing a concentrated brine solution though them, causing the above reaction to be reversed. Herein lies one of the drawbacks of this process: most of the salt employed in the regeneration process gets flushed out of the system and and is usually released into the soil or drainage system— something that can have damaging consequences to the environment, especially in arid regions. For this reason, many jurisdications prohibit such release, and require users to dispose of the spent brine at an approved site or to use a commercial service company.
Riverside County CA water softener restrictions
"Alternative" water softening methods
The great economic importance of water softening has created a large and thriving industry that utilizes a number of proven methods based on well established scientific principles. It has also unfortunately attracted a variety of operators offering technologies that are purported to be better, less expensive, easier to install, or "chemical-free", but which have never been validated scientifically and whose principles of operation are largely unexplained by the known laws of chemistry. This does not mean that such schemes cannot work (after all, we can use theory to show that under idealized conditions, water can never boil and it can never rain!), but it should inspire a good degree of skepticism. Most of the statements supporting alternative water treatement methods come from those who have a commercial interest in these devices, they are not supported by credible and independently verifiable performance data, and the explanations they offer for how they work reveal such a weak understanding of basic chemistry on the part of their authors that it is difficult to have much confidence in them.
Some dubious water-treatment processes and products
Magnetic water treatment and related pseudoscience
"Catalytic" water treatment schemes
Against this, there is some anecdotal evidence that certain magnetic and electromagnetic devices can be effective in preventing scale formation in hard water systems. It is very difficult to judge such claims, which are almost never based on tests that are well enough described to allow others to evaluate them and to verify the results. While the lack of "scientific" evidence does not in itself invalidate a claim for the efficacy of a device, it should make one hesitate to accept it without some guarantee of performance.
In 2002, an article was published in an applied physics journal that showed magnetic scale control to be effective under certain conditions, and which proposed a credible mechanism for this effect. See the MagScams page for more information.
For a quick refresher on modern science's view of water, see my
Gentle introduction to water and its structure .
This Wikipedia page covers the basics of water treatment and disinfection.
For a thorough treatment of the chemical principles relating to acid-base, carbonate, and solubility equilibria in natural waters, several text chapters from my aquatic environmental chemistry course are available in Acrobat format. Please note that these assume some knowledge of elementary Chemistry and that they do not deal directly with water treatment.
Need advice on water treatment problems? I suggest that you contact a reputable plumbing contractor who is familiar with the water in your locality. The following links may also be useful:
A very understandable consumer-oriented guide
National Science Foundation: Consumer information on
home water treatment systens, listings of certified specific water treatment products,