chem1 virtual textbook
a reference text for General Chemistry
Stephen Lower
Simon Fraser University
Trying to introduce chemical thermodynamics to beginning students is always problematic; to do it "properly" requires a degree of rigor that rarely succeeds for more than a small fraction of the class. Although a full formal development is rarely appropriate at this level, I believe that the value of developing students' understanding of the fundamental concepts is generally underappreciated. This requires some understanding of the ways thermal energy is dispersed in matter— something that is not a part of classical thermodynamics and is not supported by most textbooks, but which is in keeping with the molecular focus of modern chemical science. Although there is a lot of material in this section of the Chem1 Virtual Textbook, I believe that it is well within the intellectual grasp of first-year college students. It should be even more valuable for students in more advanced courses in physical chemistry.
Students who wonder why they are expected to learn this stuff may wish to read the introduction below.

--
To access the desired page, click one of the six lesson titles immediately below; you can also go directly to any sub-section within a lesson by clicking on a smaller heading.
1- Energy spreading drives spontaneous change
Many courses skip this stuff at the peril of turning out students who regard thermodynamics as "black magic".
- What is a spontaneous process?
- Direction through disorder
- Microstates are quantized
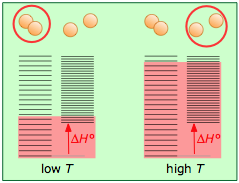
- Energy-spreading changes the world
- How chemical reactions spread energy around
- Summary of key concepts
- Concept map
2- Entropy rules! ...but what is entropy?
- Reversible and irreversible changes
- The physical meaning of entropy
- Absolute entropies
- Standard entropies of substances
- Entropy depends on concentration
- Summary of key concepts
- Concept map
3- The Second Law and the availability of energy
- Why isn't the First Law enough?
- Entropy and spontaneous change
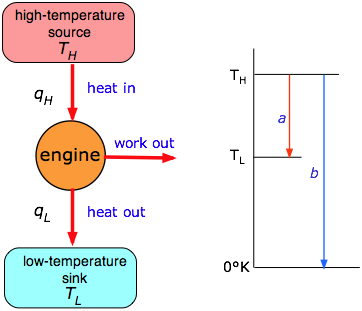
- What is a heat engine?
- The Second Law of Thermodynamics
- Summary of key concepts
- Concept map
4- Free energy and the Gibbs function
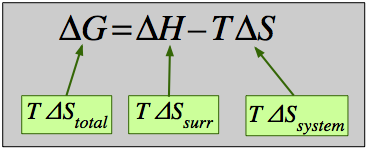
- 1 The Gibbs energy
- 2 Free energy and chemical change
- 3 The standard Gibbs free energy
- 4 ΔG and ΔG°: what's the difference?
- 5 How to interpret ΔG° values
- 6 Some remarks about "free energy"
- Summary of key concepts
- Concept map
4B- Thermodynamics of mixing and dilution
Optional topics, not normally covered in introductory courses
- 1 Review: Entropy and concentration
- 2 Gibbs energy of a gas
- 3A Chemical reactions and mixing
- 3B Dilution and diffusion
- 3C Solute standard states and activity
5- Free energy and equilibrium
- Down the free energy hill
- The equilibrium constant
- How temperature affects equilibrium
- Heterogeneous reactions
- Coupled reactions
- Summary of key concepts
- Concept map
6- Some applications of entropy and free energy
This is what makes the study of thermodynamics worthwhile!
Introduction: why this stuff is important
 The greater part of what we call chemistry is concerned with the different kinds of reactions that substances can undergo. The statement that “hydrogen fluoride is a stable molecule” is really a way of saying that the reaction HF → ½ H2 + ½ F2 has a negligible tendency to occur in the forward direction and an overwhelming tendency to occur in the reverse direction. More generally, we can predict how the composition of an arbitrary mixture of H2, F2, and HF will tend to change by comparing the values of the equilibrium constant K and the equilibrium quotient Q; in your study of equilibrium, you will recall that if Q/K > 1, the reaction will proceed to the left, whereas if Q/K < 1 it will proceed to the right. In either case, the system will undergo a change in composition until it reaches the equilibrium state where Q = K.
The greater part of what we call chemistry is concerned with the different kinds of reactions that substances can undergo. The statement that “hydrogen fluoride is a stable molecule” is really a way of saying that the reaction HF → ½ H2 + ½ F2 has a negligible tendency to occur in the forward direction and an overwhelming tendency to occur in the reverse direction. More generally, we can predict how the composition of an arbitrary mixture of H2, F2, and HF will tend to change by comparing the values of the equilibrium constant K and the equilibrium quotient Q; in your study of equilibrium, you will recall that if Q/K > 1, the reaction will proceed to the left, whereas if Q/K < 1 it will proceed to the right. In either case, the system will undergo a change in composition until it reaches the equilibrium state where Q = K.
Clearly, the value of K is the crucial quantity that characterizes a chemical reaction, but what factors govern its value? In particular, is there any way that we can predict the value of the equilibrium constant of a reaction solely from information about the products and reactants themselves, without any knowledge at all about the mechanism or other details of the reaction? The answer is yes, and this turns out to be the central purpose of chemical thermodynamics:
The purpose of thermodynamics is to predict the equilibrium composition
of a system from the properties of its components.
Don’t let the significance of this pass you by; it means that we can say with complete certainty whether or not a given change is possible, and if it is possible, to what extent it will occur— without the need to study the particular reaction in question. To a large extent, this is what makes chemistry a science, rather than a mere cataloging of facts.
Some related links:
- Chemical Energetics and the First Law of Thermodynamics
- All About Chemical Equilibrium
- Frank Lambert's pages: Shakespeare and Thermodynamics, Entropy and the Second Law, The Second Law
- Thermodynamics in a NUT-shell: all Praise the Lord Kelvin!
I am indebted to Frank Lambert's patient cajoling for setting me on the right path, and to Leonard Nash's 1971 book Chem-Thermo: A Statistical Approach to Classical Chemical Thermodynamics, which sustained me on the way.

This work is licensed under a
Creative Commons Attribution-Share Alike 3.0 License.
